European ONTOX Project: Safer Chemicals, No Animal Testing
Every year, millions of animals are used in laboratories around the world for scientific purposes, but awareness of animal welfare is rapidly growing globally. This also reflects society’s demand for more humane science. However, even though awareness of human science is increasing worldwide, unfortunately, many diseases are also on the rise. This makes society dependent on medicines, medical treatments, and many chemicals used in everyday products, as well as on scientific research.
How does this work? For example, before a new drug reaches patients, or before chemicals used in cosmetics, agriculture, or food packaging enter the market, they must be carefully assessed to ensure they are safe for human health and the environment. Many substances can cause harmful effects only after long-term or repeated exposure, which is why thorough toxicity testing is necessary. Regulations in many countries, including the EU, require that these substances be tested and confirmed as safe.
For decades, animal testing has been the standard method for assessing these risks. However, this is beginning to change.
ONTOX aims to become a turning point in chemical risk assessment for humans without using animals
In 2021, the 120th anniversary of Nobel laureate Linus Pauling’s birth was celebrated. Pauling, a chemist, was among the first to study the effects of vitamin C doses on human health. This reminds us that in toxicology, dose, concentration, and exposure are very important, and careful monitoring of exposure helps understand both safety and effectiveness.
Since his time, much has changed and been discovered. Terms such as green toxicology have emerged, aiming to reduce chemical hazards during product development by focusing on prevention rather than testing after development. Another important term is metabolomics. In the study “Metabolomics in Preclinical Safety Assessment of Drugs: Current Status and Future Trends,” researchers focused on metabolomics, a scientific approach that studies all small molecules (metabolites) in cells and tissues to better understand the effects of drugs and chemicals. Metabolomics can detect early signs of toxicity, explain mechanisms of harmful effects, and help assess risk. As the authors note: “Metabolomics is revolutionizing 21st-century toxicology through a symbiotic relationship with computational tools, microphysiological systems, and personalized medicine.” Today, artificial intelligence is present in all areas of life. AI is playing an increasingly important role in science, serving as a powerful tool that helps develop methods leading to reliable data and better decisions for the future.
All of this comes together in the European research project ONTOX (Ontology-driven and artificial intelligence-based repeated dose toxicity testing of chemicals for next generation risk assessment), coordinated by Professor Mathieu Vinken from the Vrije Universiteit Brussel and funded by the European Union through the Horizon 2020 program.

It should also be emphasized that the ONTOX project brings together a network of eighteen partners, reflecting a clear vision that removes boundaries between countries and disciplines, and, most importantly, between different interests.
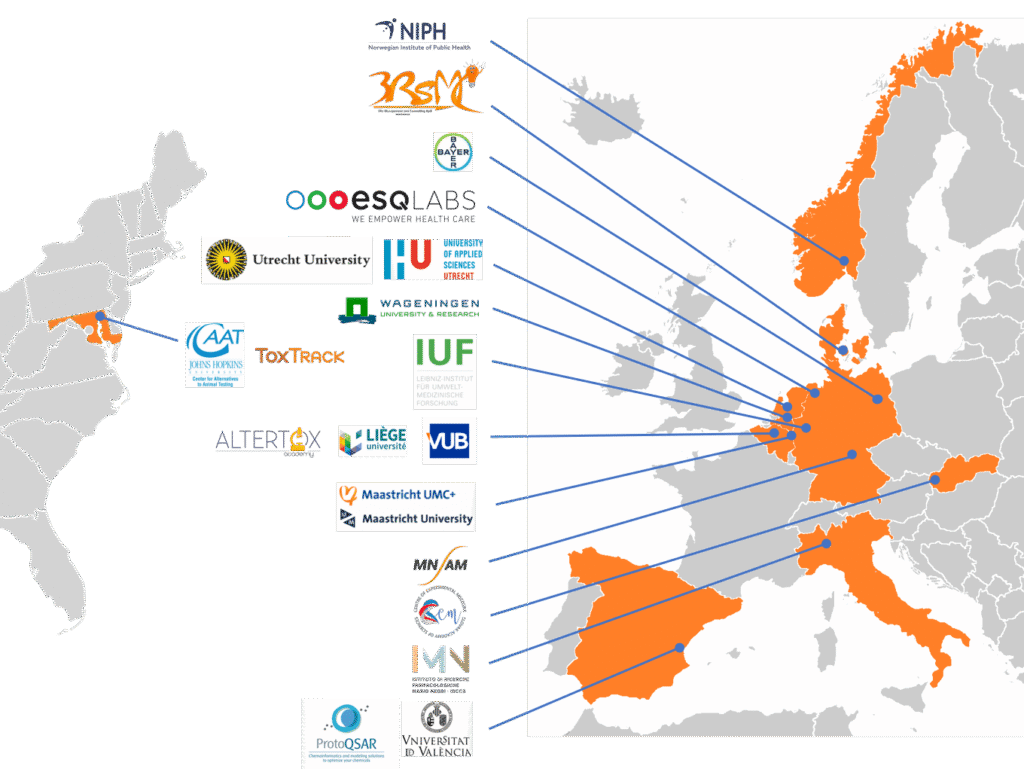
The project began in May 2021, and in the same month, a review paper was published titled “In Vitro Testing of Chemical Liver Toxicity: A Pragmatic Approach,” where researchers warned that testing liver toxicity using laboratory animals not only raises serious ethical questions but also poorly predicts chemical safety for humans. Furthermore, ONTOX is aligned with current and upcoming EU policies aimed at modernising chemical safety assessment and reducing animal testing. The project supports the objectives of REACH, the EU Strategy for Sustainability in Chemicals, and the Life Sciences Strategy, while preparing for future initiatives such as the EU Biotechnology Act and the EU plan to phase out animal testing.
In the June 2021 publication, “Safer Chemicals with Fewer Animals: Launching the European ONTOX Project,” they outlined clear ambitions and goals, from changing regulatory rules to becoming a historic turning point in chemical risk assessment for humans without animals. They emphasize: “Europe has always played a leading role globally in protecting animals used for experimental and other scientific purposes…”
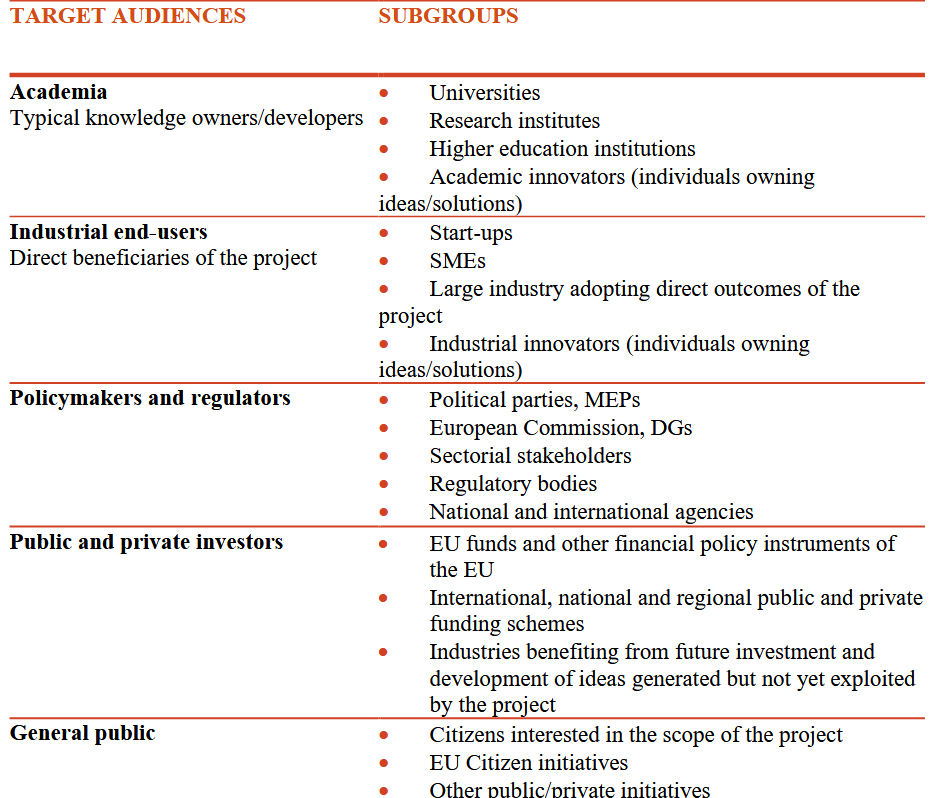
With very clear goals, strong ambition, and a solid network of partners, the ONTOX project also precisely defines its target audience, recognizing the importance of engaging society as a whole. Its work is not limited to a narrow circle of experts or politicians; it also addresses citizens, investors, policymakers…
New Approach Methodologies and the Importance of Physiological Maps
ONTOX has an ambitious goal: to predict how chemicals affect human health in the long term, without relying on animal testing. Instead of traditional approaches, the project develops New Approach Methodologies (NAMs), a sophisticated combination of biology, artificial intelligence, and big data analysis. In this approach, laboratory experiments using human cells (in vitro) and computer simulations (in silico) work together, complementing each other and filling gaps in risk assessment. The focus is on three key organs: the liver, the body’s biochemical factory that breaks down and transforms many substances; the kidneys, which act as filters maintaining internal balance; and the developing brain, which is particularly sensitive because even small disturbances early on can have long-term consequences. Instead of waiting for damage to appear, ONTOX seeks to understand the very beginning of the process, how a chemical triggers a chain of molecular and cellular changes that can eventually develop into serious health problems. These data are then integrated into computer models capable of predicting long-term health outcomes. The system is designed to expand to other organs and biological systems, making it a flexible tool for the future of toxicology.
At the heart of this approach are physiological maps, detailed representations of organ function in a healthy state. The project develops specific maps for the liver, focusing on lipid metabolism and bile secretion; for the kidneys, focusing on the complex nephron physiology; and for brain development, including neural tube closure and formation of cognitive functions. When toxicology data and exposure information are layered onto these maps, scientists can predict potential health consequences before they even start to appear. The importance of these approaches is highlighted in the study “REACH Surpassed! The Future of REACH and Analyzed Animal Numbers,” which examined how many animals are used for chemical testing under EU REACH regulations, which aim to reduce animal testing and use it only as a last resort. Results show that by the end of 2022, around 2.9 million animals were used for three main types of toxicology tests: reproductive, developmental, and repeated dose toxicity. The authors emphasize that New Approach Methodologies (NAMs) are key to reducing animal use, enabling more effective and humane testing, and modernizing chemical safety assessment in line with human and ecological standards.
Over 60 Publications Highlight Innovations in Toxicology and Drug Safety
From 2021 to the end of 2025, over 60 publications were released on the project’s website, including reports, articles, reviews, studies, and workshop reports. They cover numerous analyses, such as the study “New Clinical Phenotypes, Drug Categorization, and Outcome Prediction in Drug-Induced Cholestasis: Database Analysis of 432 Patients Using Literature Review and Machine Learning.1” In this study, the researchers collected data from 432 patients with DIC, linked to 52 different drugs. DIC, or drug-induced cholestasis, is a condition in which drugs reduce the production and flow of bile, potentially leading to bile accumulation and liver damage. The analysis showed how different drugs and their properties influence the occurrence and severity of this condition. Using the collected data, a predictive model was developed to help categorize drugs, plan therapies, and select safer candidates for new drug development.
In the publication “ToxAIcology – The Evolving Role of Artificial Intelligence in Advancing Toxicology and Modernizing Regulatory Science,” the author explained how toxicology has evolved from an observational science into a data-rich discipline that increasingly uses AI. Combining AI with high-quality scientific data can make toxicology more precise, predictable, and effective in protecting human health and the environment. The author stresses, however, that AI is not a magic solution and must be used responsibly, with transparency, ethical guidelines, and multidisciplinary collaboration.
However, although the role of AI is becoming increasingly important, nothing would be possible without mitochondria, popularly known as “the powerhouse of the cell” because of the role they play in our cells. Therefore, in the review article “Mitochondria as a Target of Hepatotoxicity and Drug-Induced Liver Injury: Molecular Mechanisms and Detection Methods,2” the authors explained the role of mitochondria in the development of drug-induced liver injury (DILI). They point out that mitochondrial toxicity is often detected too late, during clinical trials or after the drug is already on the market. Therefore, they suggest developing advanced in vitro 3D models, “organ-on-a-chip” systems, and in silico approaches to allow earlier prediction of liver toxicity and safer drug development.
How to improve strategies without relying on animals, as well as which computer models are used in the assessment of chemical hazards, can be found in “Monte Carlo Models for Subchronic Repeated Dose Toxicity: Systemic and Organ-Specific Toxicity.3“Computer models (QSAR models) for predicting the safety of chemicals for the human body are presented in the publication. More information on models that help scientists assess chemical hazards, prioritize testing, and improve safety strategies without heavy reliance on animal testing can be found on the VEGA platform.
The study, “G × E Interactions as a Basis for Toxicological Uncertainty,”4 investigated how toxic effects observed in model systems, such as animals, can be more accurately predicted in humans using the concept of gene-environment interaction. Instead of relying on standard safety factors, modeling actual variations based on genetic influence (Ge) and exposure time (Et), called G × E, is suggested. This approach helps better understand how genes and duration of exposure affect toxicity and different biological pathways of harmful effects. This method can reduce reliance on animal experiments.
From studies, reviews, and articles to reports from numerous workshops, up to the presentation of the first attempt to study DIS from a clinical perspective, as described in the study “Uncovering Drug-Induced Hepatic Steatosis: Clinical Subphenotypes, Outcome Prediction, and Identification of High-Risk Drugs and Hazardous Chemical Attributes.5” The study focused on a rare but serious type of drug-induced liver injury called drug-induced steatosis (DIS). The goal was to systematically study DIS, identify new disease subphenotypes, and link them to clinical, biochemical, and histopathological features of patients. Data on 251 cases related to 34 different drugs were collected through a literature review and using AI tools. Based on clinical and chemical characteristics, a predictive model was developed to identify high-risk drugs and dangerous chemical features. In short, the study helps understand DIS, predict risk, and potentially improve drug safety. “To our knowledge, this is the first attempt to study DIS from a clinical perspective, after building a database that includes observational clinical information from over 250 patients collected from case reports in the literature,” the authors noted.
Celebrating Achievements and Empowering Young Scientists in Humane Toxicology
The ONTOX project represents an opportunity for young scientists, offering them the chance to work on innovative chemical safety assessment methods with a strong focus on animal-free approaches. Through interdisciplinary collaboration and an international environment, early-career researchers gain scientific independence, professional confidence, and practical experience.
For example, on February 14, 2022, Dr. Eliška Kuchovská, a postdoctoral researcher at the Leibniz Research Institute for Environmental Medicine, highlighted the importance of an ethical approach in toxicology: “I am happy that European research is turning away from animal testing, which is unethical and often even makes little scientific sense—humans are simply not 80-kilogram mice.” Three years later, the experience of Julen Sanz Serrano, a postdoctoral researcher at Vrije Universiteit Brussel, confirms the positive impact of the project on the professional development of young researchers: “ONTOX truly changed and shaped me as a researcher. I became more independent and confident—no more impostor syndrome! I also developed a much broader, ‘helicopter view’ of my work and learned everything I know about risk assessment…”
The team’s achievements have also been recognized through awards. In late September 2024, Prof. Mathieu Vinken, coordinator of the ONTOX and ASPIS projects, received the Dr. Monique Govaerts and Joseph Lepoix Award for predicting human chemical safety using AI and in vitro methods without animal testing. In late 2025, ONTOX further strengthened international collaboration by participating in the ChemBAI 2025 symposium at Seoul National University, while in January 2026, Prof. Vinken was awarded the prestigious 2026 SOT Enhancement of Animal Welfare Award for advancing human-relevant toxicology and effective alternatives to traditional animal testing.
In addition, ONTOX collaborates with other European projects, such as PARC, NAMwise, VICT3R, and VHP4Safety, further strengthening the European scientific base through knowledge and experience exchange.
What ONTOX Means for Us: A New Era of Humane and Sustainable Toxicology with Fast Data Collection
ONTOX aims to represent a turning point in toxicology, marking a transformative shift toward chemical safety assessment that is simultaneously more ethical, precise, and human-relevant. They also work on developing tools, as seen in the study “ExtractPDF: A Tool for Extracting Data from Scientific Papers Applied to Systematic Reviews in Public Health.6” This study introduced the ExtractPDF tool, designed to automate the extraction of information from unstructured PDF files. This tool can help accelerate the data collection phase in systematic reviews and ensure the validation of extracted information. ExtractPDF allows reliable extraction of words or word combinations from a large number of PDF documents. It was applied to 299 scientific papers within a systematic review in public health risk assessment.
In the study “Classification of Steatogenic Compounds Using Machine Learning with Toxicogenomic Profiles,7” the application of machine learning was presented to predict drug-induced fatty liver disease using gene expression data from human and rat liver models. The support vector machine (SVM) algorithm showed the highest predictive accuracy among the tested models. Results show that machine learning can identify early molecular signs of disease, providing human-relevant information for chemical safety assessment without using animals. “To enable real-world implementation, active participation of regulatory stakeholders will be crucial for defining validation criteria and ensuring that omics-based machine learning models can be properly assessed and integrated into regulatory frameworks.”
Traditional testing often involves thousands of animals, and due to biological differences, results are not always applicable to humans. By reducing or replacing animal tests with data-driven and AI-based methods, ONTOX enables scientific progress without harming living beings. For humans, the project provides more accurate and reliable chemical risk assessments. Using NAMs and AI-driven models allows detailed analysis of chemical exposure, organ-specific toxicity, and long-term health outcomes. For the environment, the project also contributes significantly to its protection and sustainable development. By minimizing animal testing, the project reduces the ecological footprint of laboratory research, including energy, water use, and lab waste. For future generations, this is an example of how innovation can be morally responsible. Children and young people exposed to these approaches learn the importance of empathy toward animals and the environment. The project raises awareness that progress should not be heartless, but should include ethical and sustainable principles.
ONTOX serves as a powerful reminder not only of how much we have advanced over the past 120 years but also that progress and innovation should not require a compromise between scientific excellence and moral responsibility. This project clearly demonstrates that the toxicology of the future can be ethical, sustainable, and scientifically rigorous, protecting animals, humans, and the environment, and that such a future has never been closer.
Image: ONTOX
1.This study has been funded by the European Commission under the Horizon2020 Research and Innovation Framework program (grant number 963845 “ONTOX”) and by Instituto de Salud Carlos III (ISCIII) through the project “PI20/00690” (to RJ) and co-funded by the European Union. MMT acknowledges the grant RYC2021–031346-I, funded by MICIU/AEI/10.13039/501100011033 and by the European Union NextGenerationEU’/PRTR and the project CIGE/2022/145 from the Conselleria de Educación, Universidades y Empleo (Generalitat Valenciana). GQ acknowledges the grant PID2021–125573OB-I00 funded by MICIU/AEI/10.13039/501100011033 and by ERDF A way of making Europe, by the “European Union”.
2.This work was financially supported by the Marie Skłodowska-Curie COFUND Action (Grant Agreement 101034352 “imπACT”), the Future and Emerging Technologies (Grant Agreement 858014 “PANACHE”) and the Societal Challenges (Grant Agreement 963845 “ONTOX”) sections of the European Horizon2020 framework program, the US Alternatives Research and Development Foundation (Grant 2021), the Research Foundation Flanders (Grant Agreement G070422N), the Methusalem program of the Flemish government (Grant 2020), and the Scientific Fund Willy Gepts (Grant 2021).
3.This research was funded by the European Union’s Horizon 2020 research and innovation programme under Grant Agreement No. 963845 (ONTOX).
4. Open Access funding enabled and organized by Projekt DEAL.
5. This study has been funded by the European Commission under the Horizon2020 Research and Innovation Framework program (grant number 963845 “ONTOX”), by Instituto de Salud Carlos III (ISCIII) through the project “PI24/00215” (to MMT and RJ) and co-funded by the European Union, and by Grant CNS2024–154299 funded by MICIU/AEI /10.13039/501100011033 (to MMT). MMT acknowledges the grant RYC2021–031346-I, funded by MICIU/AEI/10.13039/501100011033 and by the European Union NextGenerationEU/PRTR.
6. This project received funding from the European Union’s Horizon 2020 Research and Innovation programme under Grant Agreement No 963,845 (ONTOX project).
7.This work was performed in the context of the ONTOX project (https://ontox-project.eu/) that has received funding from the European Union’s Horizon 2020 Research and Innovation programme under grant agreement No 963845. ONTOX is part of the ASPIS project cluster (https://aspis-cluster.eu/). Additional support for this work was provided by the Virtual Human Platform for Safety Assessment (VHP4Safety) project, which is funded by the Netherlands Research Council (NWO) Netherlands Research Agenda: Research on Routes by Consortia (NWA-ORC 1292.19.272).
These publications were produced as part of the Maria Leptin EMBO Fellowship, which allowed us to spend two months exploring the world of science at VUB in Brussels. Importantly, all articles were the result of our own choice of topics and in accordance with our interests.


