KARMA-Dep 2: Ketamine Under Review
During electroconvulsive therapy (ECT), short electrical impulses are passed through the brain to trigger a controlled seizure, which can lead to significant improvement in symptoms of severe mental illnesses such as depression. The procedure is performed under general anesthesia.
A study that analyzed data from three U.S. states over a period of up to 17 years and included 62,602 patients found that the average recipient of ECT was more likely to be female and older. The researchers noted that these trends persisted throughout the study period, which spanned from 2001 to 2018.
About 30% of patients do not respond adequately to antidepressants
When it comes to depression, about 30% of patients do not respond adequately to antidepressants, which is why, as scientists note, new treatments are needed. In recent years, ketamine has gained attention as a potential alternative to electroconvulsive therapy (ECT) for patients with severe depression. It has shown rapid antidepressant effects in single infusions, but they disappear after just a few days. It is generally considered safe, though not without risk. According to the study, “there are reasons to believe that ketamine has addictive properties with risk for tolerance and dependency.”
The new study aimed to assess whether multiple infusions could maintain these effects. The results were published in a study titled, “Serial Ketamine Infusions as Adjunctive Therapy to Inpatient Care for Depression: The KARMA-Dep 2 Randomized Clinical Trial.”
We spoke with Declan McLoughlin, who led the KARMA-Dep (2) Trial, which involved researchers from St Patrick’s Mental Health Services, Trinity College Dublin, and Queen’s University Belfast, Ireland. McLoughlin is Research Professor of Psychiatry at Trinity College Dublin and Consultant Psychiatrist at St Patrick’s Mental Health Services.
The KARMA-Dep 2 Randomized Clinical Trial
The study involved 62 randomized participants in a blinded clinical trial (they did not know which treatment they had been allocated) who received up to eight intravenous infusions of either ketamine or midazolam, administered twice weekly, in addition to usual psychiatric care. Participants were followed for six months. Before the trial, electronic medical records of all hospital admissions (slightly more than 3,500 admissions) were reviewed. Those who were potentially eligible (371 patients) were invited to a screening visit, and if they agreed ( 74), they provided written informed consent.
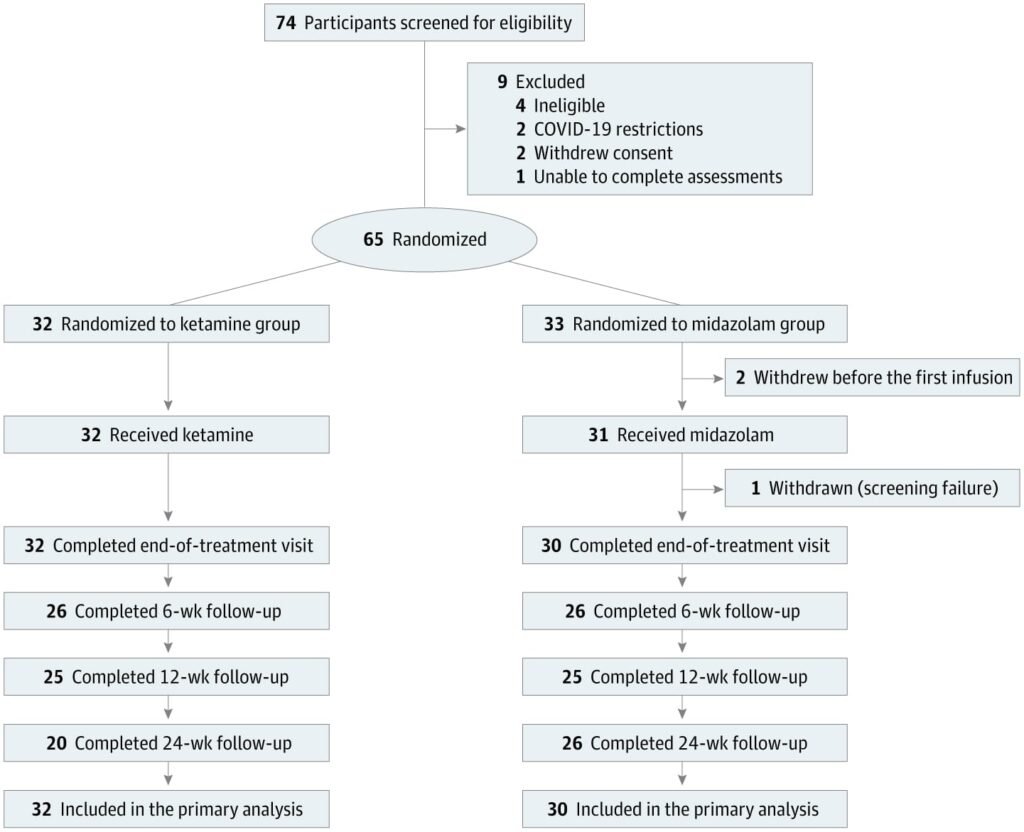
Results did not differ significantly between the ketamine and midazolam groups, meaning that ketamine infusions were not more effective than serial midazolam infusions in reducing depressive symptoms. The study also showed that the vast majority of patients and raters correctly guessed the treatment allocation, which could have led to enhanced placebo effects.
According to the study, mean costs from baseline to 24-week follow-up were €6553.54 (US $7724.63) for ketamine (n = 32) and €6154.91 (US $7254.76) for midazolam (n = 30). The trial was conducted between September 13, 2021, and August 12, 2024, at St Patrick’s University Hospital in Dublin, Ireland.
A real risk of patients and clinicians being steered towards a less effective treatment
What are the implications of these findings for future research, and how can they be applied in clinical practice?
Professor McLoughlin: Our initial hypothesis was that repeated ketamine infusions for people hospitalised with depression would improve mood outcomes. However, in our KARMA-Dep 2 trial, we found this not to be the case. Under rigorous clinical trial conditions, adjunctive ketamine provided no additional benefit to routine inpatient care during the initial treatment phase or the six-month follow-up period. Previous estimates of ketamine’s antidepressant efficacy may have been overstated, highlighting the need for recalibrated expectations in clinical practice.
What do you think is the biggest challenge in this case, especially considering the increasing number of people affected by depression?
Professor McLoughlin: Our trial highlights the importance of reporting the success, or lack thereof, of blinding in clinical trials. Especially in clinical trials of therapies where maintaining the blind is quite difficult, e.g., ketamine, psychedelics, and brain stimulation therapies. Such problems with blinding can lead to enhanced placebo effects and skewed trial results that can over-inflate real treatment effects.
There is no simple answer to this question, and it’s a major unresolved challenge in designing ketamine and psychedelic trials going forward. To some extent, in our own trial, we addressed this by recruiting only people who had been admitted to the hospital for depression. We did not advertise the trial and did not hype any expectations. So, our trial participants were not people seeking to participate in ketamine research. This may have helped reduce expectancy bias.
“Studies show that about 30% of people with depression do not respond adequately to conventional antidepressants, which mostly target monoamine neurotransmitters such as serotonin, dopamine, and noradrenaline. There is therefore a need for new treatments.” How can we help these patients, and what do you think needs to change in our approach?
Professor McLoughlin: This is indeed a major clinical problem. It is possibly compounded by too readily diagnosing people as having a depressive illness when this may well be something else, such as an adjustment disorder. The clinical situation can be further complicated by comorbid personality difficulties, anxiety disorders, substance misuse, etc. In such instances, treatments for depression may not be of much use. This can probably be decreased by directing people towards more relevant therapies and psychosocial supports.
On a separate but related point, ketamine has recently been suggested to be an alternative to electroconvulsive therapy (ECT) for depression. However, scrutiny of the scientific literature shows that this includes methodologically flawed trials and invalid meta-analyses. We discuss this in some detail in a Comment piece just published in The Lancet Psychiatry. Unfortunately, such errors have been accepted as scientific evidence and are already creeping into international clinical guidelines. There is thus a real risk of patients and clinicians being steered towards a less effective treatment, particularly for patients with severe, sometimes life-threatening, depression.
As scientists point out in a commentary published in The Lancet, the claim that ketamine is equivalent to ECT is also not supported by credible evidence. They warn that patient care must be guided by solid evidence, not misinformation or errors in clinical practice and public policy.
This work was supported by the Health Research Board of Ireland (grant DIFA-2018-013).


